



Ion exchange reactions
Ion exchange reactions - эit's such a chemical reaction, substances in which the exchanged ions.
For example, in the interaction of copper sulphate and sodium hydroxide (alkali), it forms a gelatinous sediment blue:
CuSO4+2NaOH=Na2SO4+Cu(OH)2
Here is an exchange of ions of copper and sodium. A blue solution of copper sulfate CuSO4, reacting with alkali NaOH, discolored and fall out blue gelatinous sediment of copper hydroxide Cu(OH)2 and is forming light solution is a sodium sulfate Na2SO4.
Similarly, reaction of the Nickel sulfate Ni2SO4 and caustic soda NaOH. Only instead of a blue sediment is a green nickel hydroxide Ni(OH)2
NiSO4+2NaOH=Na2SO4+Ni(OH)2
Beautiful reaction of ion exchange happens if solution of ammonia) add to a blue solution of copper sulfate CuSO4. When mixing these solutions ammonia adds a copper ion and forming a complex compound ammicht copper is a beautiful rich blue solution.
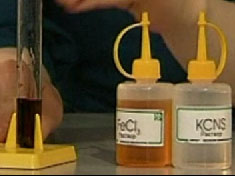
The following chemical reaction ion exchange is high quality chemical reaction ion III-valent iron. If you pour a solution of ferric chloride FeCl3 to a solution of potassium thiocyanate KCNS, the solution will change its color to blood red. This forms a complex compound - iron thiocyanate Fe(CNS)3.
Reaction neutralization (or neutralize acid)
To chemical neutralization reactions are reactions between acid and alkali (base), which is formed of water and some salt.
An example of such a process may be a reaction between the hydrochloric acid HCl and sodium hydroxide NaOH
HCl+NaOH=NaCl+H2O
Salt formed in reaction - sodium chloride - solution of salt in water (but still, trying not recommended!). Neutralization of acid and alkali can be observed, if at the beginning of the acid solution (for special effect!) add a few drops of some indicator - to change the color of the solution. And when the resulting colored solution (acid indicator) to add alkali, the color will disappear - it looks like a miracle! You can use any acid or any alkali: for example, citric acid or ammonia (ammonium hydroxide NH4OH).
Hydrolysis
The word hydrolysis means the decomposition substances with water, parts of which after decomposition connected with parts of water. On the other you can say that it is the interaction with water, result is formation electrolyte with pH environment.
For example, hydrolysis of salts are exchange reactions with parts of water. You can still say that hydrolysis is the reverse reaction neutralization, which forms a weak acid or weak base.
We consider the hydrolysis, for example, one of the most common salt used in the household. Remember what is the salt! - this is a complex substance that is similar with acid, in which the atom (or atoms) of hydrogen is replaced by an atom (or atoms) metal (HCl - hydrochloric acid, and NaCl (salt).
Now, consider the hydrolysis for example, washing soda:
Washing soda - sodium carbonate (chemical formula Na2CO3 - her second name soda ash) is perfectly soluble in water, i.e. interacts with it, and then subjected to hydrolysis.
Washing soda is formed of a weak acid ( carbonic acid (H2CO3) and strong base (sodium hydroxide NaOH).
That's what happens when you dissolve this salt in water (soda in water):
Soda dissociates (breaks apart) ions:
2Na++CO32-+H++OH- = Na++HCO3-+Na++OH- - 1st stage (full ionic form)
Na++HCO3-+H++OH- = H2CO3+Na++OH- - 2nd stage (full ionic form)
In molecular form can be written:
Na2CO3 + H2O = NaHCO3 + NaOH - 1st stage
NaHCO3 + H2O = H2CO3 + NaOH - 2nd stage
As you can see, the dissociation form free ions OH-, which indicate the presence a strong base (NaOH) in solution, and solution is strongly alkaline (for washing in such water needs gloves).
Similarly, if dissolved in water hydrolysis, for example, copper chloride (CuCl2) - but this salt is formed by a weak base (copper hydroxide Cu(OH)2) and a strong acid (HCl - hydrochloric acid), and the result of hydrolysis will produce a solution with a strong acidity. Sometimes in you can see in books that the water (H+ OH-) is a substance that weakly dissociates to ions
.