Properties of oxides
Oxides is a complex chemical substances that represent simple chemical compounds of the elements with oxygen. They are salt-forming and do not form salts.
There are 3 types salt-forming oxides: Basic oxides (from the word "Base"), acidic oxides and Amphoteric oxides.
Example of oxides, not forming a salt may be: NO (nitric oxide) is a colorless gas, odorless. It is formed by electrical storms in the atmosphere. CO (carbon monoxide) gas is odourless, is formed by the combustion of coal. It is usually called carbon monoxide.
There are other oxides, that do not form salts.
Now let consider each type of salt-forming oxides.
Basic oxides
Basic oxides -it is a complex chemical substance oxides, which form a salt with the chemical reaction with acids or acidic oxides and do not react with bases or basic oxides. For example, the basic oxides include the following:
K2O (potassium oxide), CaO (calcium oxide), FeO (iron oxide 2-valent).
Consider the chemical properties of oxides examples
1. Reaction with water:
- react with water and form of the base (or alkali)
CaO+H2O → Ca(OH)2 (reaction as known as the liming, it releases a large amount of heat!)
2. react with acids:
the react with acid and form of salt and water (a solution of salt in water)
CaO+H2SO4 → CaSO4+ H2O (Crystals of this substance CaSO4 everywhere known as "plaster of Paris").
3. react with acidic oxides: form salts
CaO+CO2→CaCO3 (This substance as known as chalk!)
Acidic oxides
Acid oxides is a complex chemical substance oxides, which form a salt with the chemical reactions with bases or basic oxides and do not react with acidic oxides.
Examples of acidic oxides can be:
CO2 (all known carbon dioxide), P2O5 - oxide of phosphorus (formed in air if burns white phosphorus), SO3 - oxide of sulfur (VI) is a substance used for sulfuric acid
- chemical reaction with water
CO2+H2O→H2CO3 is a substance - carbonic acid is one of the weak acids, it is added to carbonated water for "bubbles" of gas. With increasing temperature the solubility of gas in water decreases, and the excess comes out in the form of bubbles.
- reaction with alkalis (bases):
CO2+2NaOH→Na2CO3 +H2O - formed substance (salt) is widely used in agriculture. It's called soda ash or washing soda, is a great cleanser for burnt pans, fat, burn-on. Bare hands to work I do not recommend!
- the reaction with the basic oxides:
CO2+MgO→MgCO3 is obtained salt is magnesium carbonate is also called "bitter salt".
Amphoteric oxides
Amphoteric oxides - it is a complex chemical substance, also oxides, which form a salt with the chemical reactions with acids (or acid oxides) and with bases (or basic oxides). The most frequent used the word "amphoteric" for the oxides of metals.
Example amphoteric oxides can be:
ZnO - zinc oxide (white powder, often used in medicine to produce masks and creams), Al2O3 - aluminum oxide (also called "alumina").
Chemical properties of amphoteric oxides are unique in that they can enter into a chemical reaction, appropriate as bases and acids. For example:
- reaction with acidic oxide:
ZnO+H2CO3 → ZnCO3 + H2O - Formed substance, the composition of the salt of zinc carbonate in water.
- reaction basis:
ZnO+2NaOH→Na2ZnO2+H2O - obtained substance is a double salt of sodium and zinc.
Getting oxides
Getting oxides produced in various ways. This can occur by physical and chemical methods. The easiest way is chemical reaction the simple elements with oxygen. For example, the result of the combustion process the one of the products of this chemical reaction are oxides.
For example, if a red-hot iron rods (you can take zinc Zn, tin Sn, lead Pb, copper Cu, is actually that you have) is placed in the flask with oxygen, there will be a chemical reaction oxidation, which is accompanied by a bright flash and sparks. The reaction product is a black powder of iron oxide FeO (For example, if it was iron):
2Fe+O2→2FeO
Completely analogous the chemical reactions of oxidation for other metals and nonmetals, Such as:
Zinc burns in oxygen to form zinc oxide
2Zn+O2→2ZnO
Burning coal is forming two oxides: carbon monoxide and carbon dioxide
2C+O2→2CO - formation of carbon monoxide.
C+O2→CO2 is the formation of carbon dioxide. This gas is formed when oxygen is available in more than sufficient quantities, that is, in any case, the first reaction takes place with the formation of carbon monoxide, and then carbon monoxide is oxidized into carbon dioxide.
Getting oxides can be done in another way - by chemical decomposition reaction. For example, to obtain iron oxide or aluminum oxide you must ignited appropriate basis of these metals in fire
Fe(OH)2→ FeO+H2O



2Al(OH)3→ Al2O3+3H2O,
as well as the decomposition of individual acids:
H2CO3→ H2O+CO2 - decomposition of carbonic acid
H2SO3→ H2O+SO2 - decomposition of sulfurous acid
Production of oxides can be carried out from metal salts under strong heating:
CaCO3→ CaO+CO2 - igniting chalk receive calcium oxide (or lime) and carbon dioxide.
2Cu(NO3)2→ 2CuO + 4NO2 + O2 - in this decomposition reaction is obtained two oxide: copper CuO (black color) and nitrogen NO2 (it is also called brown gas because its really brown).
Another way you can make oxides with redox reaction, for example
Cu + 4HNO3(конц.)→ Cu(NO3)2 + 2NO2 + 2H2O
S + 2H2SO4(conc.)→ 3SO2 + 2H2O
Oxides of chlorine

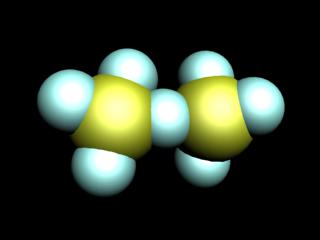
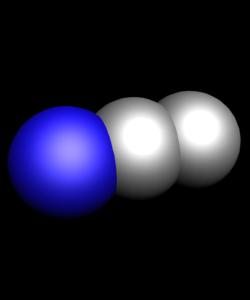
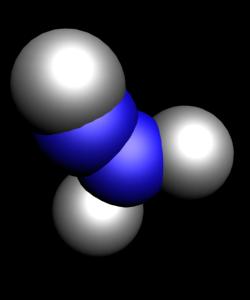
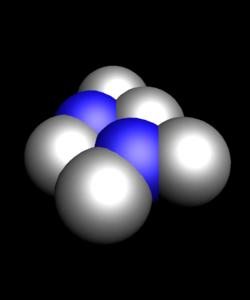
Known oxides of chlorine: Cl2O, ClO2, Cl2O6, Cl2O7. All of them, except for Cl2O7 have a yellow or orange color and not sustainable, especially ClO2 Cl2O6. All oxides of chlorine explosive and are very strong oxidants.
Reacting with water, they form a corresponding oxygen-containing and chlorine acids:
So, Cl2O - acid chlorine oxide of hypochlorous acid.
Cl2O + H2O → 2HClO - Hypochlorous acid
ClO2 - acid chlorine oxide of hypochlorous and charnawati acid as the chemical reaction with water forms two acids:
ClO2 + H2O → HClO2 + HClO3
Cl2O6 is also acid chlorine oxide of charnawati and perchloric acid:
Cl2O6 + H2O → HClO3 + HClO4
Finally, Cl2O7 is a colorless liquid acid chlorine oxide of chloric acid:
Cl2O6 + H2O→ HClO3 + HClO4
and, Cl2O7 - colorless liquid - acidic chlorine oxide perchloric acid:
Cl2O7 + H2O→ 2HClO4
Nitrogen oxides
Nitrogen is a gas that forms a 5 various compounds with oxygen - 5 oxides. Such as:
- N2O - remixed nitrogen. It has other name, is known in medicine as laughing gas or nitrous oxide gas is colourless, sweet and pleasant to the taste. NO nitrogen monoxide is a colourless, with no smell or taste the gas.
- N2O3 - nitrous anhydride is a colourless crystalline substance
- NO2 - nitrogen dioxide. Its other name is brown gas - gas really has rusty-brown color
- N2O5 - nitrogen anhydride blue liquid, boiling at a temperature 3,5 0C
Out of all these listed compounds of nitrogen of greatest interest in the industry are NO - nitrogen monoxide and NO2 is nitrogen dioxide. nitrogen Monoxide (NO) nitrous oxide N2O doesn't react with water or alkali. Nitrous anhydride (N2O3) by the reaction with water forms a weak and unstable nitrous acid, HNO2, which in the air is gradually transformed into a more stable chemical substance nitric acid
Consider some of the chemical properties of nitrogen oxides: Reaction with water:
2NO2 + H2O → HNO3 + HNO2 are formed 2 acids: nitric acid HNO3 and nitrous acid.
Reaction with alkali:
2NO2 + 2NaOH → NaNO3 + NaNO2 + H2O - formation of two salt: sodium nitrate NaNO3 and sodium nitrite
Reaction with salts:
2NO2 + Na2CO3 → NaNO3 + NaNO2 + CO2 - formed two salt: sodium nitrate and sodium nitrite, and carbon dioxide.
Get nitrogen dioxide (NO2) from nitrogen monoxide (NO) by chemical reaction of compound c oxygen:
2NO + O2→ 2NO2
Iron oxides
Iron forms two oxide: FeO - iron oxide (2-valent) - black powder, which is obtained by restoring the iron oxide (3-valent) carbon monoxide according to the following chemical reaction:
Fe2O3+CO → 2FeO+CO2
This basic oxide, easily entering into reaction with acids. It has restorative properties and is rapidly oxidized to iron oxide (3-valent).
4FeO +O2 → 2Fe2O3
Iron oxides (3-valent) - red-brown powder (hematite), that has amphoteric properties (react with acids and alkalis). But the acid properties of this oxide is expressed so poorly that most often it is used as basic oxide.
There are also mixed iron oxide Fe3O4. It is formed during combustion of iron, a good conductor of electric current and has magnetic properties (it is called magnetic hematite or magnetite).
If iron is burned, the reaction form two oxides: iron oxide (III) and (II) valence.
Sulfur dioxide

Sulfur dioxide SO2 - or sulphurous gas is acid oxides, but it does not form acid, although perfectly soluble in water - 40L oxide sulfur in 1 l of water (for convenience of drafting chemical equations such solution is called sulphuric acid).
At normal, it is a colourless gas with a pungent and suffocating smell of burning sulfur. When the temperatures is -10 0C it can be changed into a liquid state.
In the presence of a catalyst of vanadium oxide (V2O5) sulfur dioxide attaches the oxygen and turns into sulfur trioxide
2SO2 +O2 → 2SO3
Dissolved in water sulfur dioxide - SO2 is very slow oxidized, and resulting is turned into sulfuric acid
If sulfur dioxide pass through a solution of alkali, such as sodium hydroxide, then a sodium sulfite (or hydrosulfite, it is depends on how much you take of alkali and sulfur dioxide)
NaOH + SO2 → 2NaHSO3 - sulfur dioxide taken more than it needs
2NaOH + SO2 → Na2SO3 + H2O
If sulfur dioxide does not react with water, then why its aqueous solution gives an acidic reaction?! Yes, not react, but he himself is oxidized in the water and adds the oxygen. And it turns out that hydrogen atoms accumulate in the water, which give an acid reaction (you can check it of some indicator!)