Metal alloys

When alloying pure metals the change their structure: the lattice sites are either closer together or moved apart. This creates additional stress, tthat creates additional forces between the atoms in the metal. That is why metal alloys is much stronger and harder than pure metals. Alloys have magnetic and mechanical properties, that was not pure metals. For example, the best permanent magnets made from alloys of magnesium, nickel and cobalt, from aluminium, nickel and cobalt or cobalt alloy with rare earth metal samarium (Sm).
Cast iron is an alloy of pure iron with carbon, in which carbon from 2.14% to 4 %. Cast iron harder than iron, but more brittle, has excellent casting properties (much better than pure iron).
Steel is also an alloy of iron with carbon, but carbon is here to 2.14%. From steel (as pure iron) is made of a pipe, bots, nails, staples, tool. There are a large number of varieties of steels, whose composition in addition to carbon include other pure metals (alloying elements), giving them different strength properties.
The most common nonferrous alloys include:
bronze - alloy based on copper with the addition up to 20% of tin;
brass - copper alloy containing from 10 to 50% of zinc;
Melchior is an alloy of 80% of copper and 20% of Nickel;
aluminum is an alloy based on aluminum with the addition of 2% of magnesium, 2-5 % of copper and 1% manganese and Nickel.
Solid solutions
When cooling the aqueous solutions of them can crystallize solid solutions. Their nature is different and depends on the components that make up a solid solution. The formed solid solutions are composed of crystals with ions atoms, molecules in the sites of the crystal lattices, that comprise a solid solution.
If the size and shape of the crystal lattices of the constituent components of a close, these components may dissolve in each other in unlimited quantities and to form solid solutions.
Types of solid solutions
With the help of x-ray studies found that solid solutions are divided into 2 types:
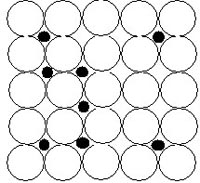
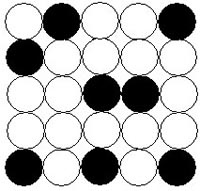
- Solid solution introduction
- Solid solution of substitution
In solid solutions of substitution of ions or atoms of one substance replaces the ions or atoms in the crystal lattice of another substance without significantly changing the shape of the crystal. There are isomorphic solid solutions of substitution, that is consisting of simple substances, similar to the parameters of the crystal lattice and have unlimited solubility in each other.
Solid solutions of introduction - are formed when atoms or ions of one substance are embedded (or placed between) the atoms or ions of another substance.
Properties of solid solutionsProperties of solid solutions differ significantly from the properties of the components. Solid solutions are technically more valuable than the pure components. They have higher hardness, lower electrical conductivity than the metals - their components.
In solid solutions have solvent and solute: solvent is the substance that save its crystal lattice, and this substance (solvent) must be at least a certain value.