
Rust - is one of the complex substance of iron formed with chemical reaction water and iron. Already at a humidity of 50 % the surface of iron is covered with a layer of water thickness of 15 molecules. Corrosion enhanced with the presence of ions Cl-, which transferred the rust in the solution to the form of [Fe(H2O)2Cl4]. As the corrosion inhibitors use a solution of NaNO2, in glycerin, amines (buty- amine) and its salts.
In air Fe(OH)2 is rapidly oxidized, turning into a yellow-brown iron hydroxide(III). Unlike Fe(OH)2, it is amphotere, although its acidic properties are much weaker than base properties; it dissolves only in concentrated alkali when heated:
Fe(OH)3+3КОН→ K3[Fe(OH)6].
The figure on the left - oxidation of Fe(OH)2 in air. A white precipitate of Fe(OH)2 (at the bottom of the tube) immediately oxidizes to yellow-brown Fe(OH)3 on the air.
The intermediate oxidation product is a dirty-green precipitate of the hydroxide containing Fe (II) and Fe (III).
Interesting, but unpleasant feature of rust is it has greater volume than the original iron. It is can cause disastrous consequences: although the rust seems to be soft, and with its growth develop huge efforts. When in London by sir Christopher in 1675-1710 built grandiose St Paul's Cathedral, the stone blocks of the bell tower were joined with iron clamps, which were laid in the grooves carved into the stone. For hundreds of years, staples have rusted and began to raise a stone laying, then the bell tower was warped. Engineers have calculated the pressure is developed with rust so great, that even would be it lifted a two-kilometer layer of stones! It needs to disassemble the clutch and insert into the grooves new steel brackets.
The rate of iron oxidation greatly depends on the total surface of contact between metal and air. So, nail, even if it is very hot, not soon turn into dross. Small sawdust then its strong heat, burn quickly, and if its put to flames flare, then its up in the form of sparks. The chemical way is the reduction of iron oxides with hydrogen — you can get more fine powder of iron; it is called pyrophoric. It flashes in the air at normal temperatures. Other metals can be pyrophoric if its have powder condition and the oxide FeO.
Corrosion of metals - (from the Latin corrodere - is chew) the processes, that occur as a result of chemical exposure of environment, and result is destruction of metal.
From this process is another process - erosion of metals, representing the destruction of metal surface under mechanical influence of the environment.
Corrosion of metals means dissolution, that begins to surface. As a result of chemical interaction with the environment. This process is spontaneous, and is a consequence of redox reaction with components of the environment. As a result of destruction of metal formed products of its oxidation, namely, oxides, hydroxides, sometimes it just happens dissolution in the environment to the ionic state. This transformation is accompanied by a significant change in properties.
There are various types of corrosion. One of its species is chemical corrosion, which is also sometimes called gas corrosion, as sometimes it happens under the influence of gaseous components from the environment at high temperatures. Chemical corrosion can occur under the influence of some corrosive liquids. "Key" to this process is that it occurs without the occurrence in the system of the electric current. Parts and units of machines, working in an atmosphere of oxygen at high temperatures, such as turbine engines, rocket engines and some others exposed it and also the details of the units of equipment of chemical production.
Another common type of destruction of metals is electrochemical corrosion - surface destruction in the environment of the electrolyte with the emergence of the system of the electric current. Electrochemical corrosion is the destruction in the atmosphere, soil, water bodies, soils.
The nature of the destruction surface of the metal may be different and depends on the properties of this metal and the conditions of the process. Now to learn more about electrochemical corrosion.
Types of corrosion
Rust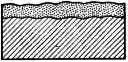




The main types of corrosion include:
Electrochemical corrosion is more intensive if the cathode is impregnated metal, less active than corrosive. For example, if corrode steel (steel is an alloy of iron and carbon in which the partially formed iron carbide) role of such sites is iron carbide(FeC).
Atmospheric corrosion
- occurs in moist air at ordinary temperature. The metal surface is covered with a film of moisture containing dissolved oxygen. The intensity of the destruction of the metal increases with increasing air humidity and gaseous oxides of carbon, sulfur, in the presence of the metal bumps, cracks facilitate condensation.
Soil corrosion
- it exposed pipelines, cables, underground structures. In this case, the metals in contact with soil moisture containing dissolved oxygen. In wet soil, with high acidity pipelines are destroyed within six months after they are laid (of course, if you don't take measures to protect them).
Electric corrosion
- occurs under the action of stray currents arising from extraneous sources (power lines,electric Railways, and various electrical installations operating at a constant electric current). Stray currents cause the destruction of pipelines, oil pipelines,electrical cables, various structures. Under the action of electric current on ground-based metal items there are areas of the entrance and exit of electrons - cathodes and anodes. The most intense destruction is observed at anodic sites.
Electrochemical corrosion
Electrochemical corrosion - the most common type of destruction of metals. An example of electrochemical corrosion is the destruction of parts of machines, devices and various metal structures in soil, groundwater, river water and sea water, in the atmosphere under the film of moisture, in the technical solutions under the action of lubricant-coolant, etc. As already mentioned, electrochemical corrosion flows on the surface of metals under the action of electric currents, that is to happen a redox chemical reaction, characterized by the faile of electrons and transfer them as generate cathodic and anodic areas. The formation of cathodes and anodes contribute to the chemical heterogeneity of metals (impurities and inclusions), the presence of areas of residual deformation, the heterogeneity of the covering metal protective films, etc. Most often in the formation of type of destruction of the metal involved is not one factor, but several. When the metal begins to destruct, it turns into a many-electron galvanic element.
For example, consider what happens if copper (Cu) is in contact with iron (Fe) in the electrolyte. This system is a galvanic element, where iron is the anode ( " + " ) and copper - cathode. Iron gives electrons to copper and goes into solution as ions. Hydrogen ions move to copper, where discharged. The cathode is gradually becoming more negative, in the end, becomes equal to the potential of the anode and corrosion slows down.
Just on this topic you can spend the experience at home. We will need three cups with a solution of sodium chloride (table salt), 3 iron nail, a piece of zinc and copper wire (without insulation). So, let's start. The first nail drop into the glass with the salt solution. The second nail with screw copper wire drop into the next glass with the salt solution, and a piece of zinc drop into the last glass. Then dip each nail into your glass with salt solution (there were 3) and leave them for 2-3 days.
What happens: all of our nails will have traces of rust (corrosion). In the worst condition will be the nail that was in the solution together with copper wire, and the least corroded one that is bound to the zinc! Explanation: all metals have different ability to give up electrons. To compare them in this property by reading number of stresses metals:
Li← K← Rb← Cs← Ba← Ca← Na← Mg← Al← Mn← Cr← Zn← Fe← Cd← Co← Ni← Sn← Pb← H2← Cu← Ag← Hg← Pt← Au
Those metals that are in the row voltages are to the left (e.g. Zn - zinc is to the left of the Fe - iron), it is easier to give their electrons than the metal to the right (for example Cu - copper to the right of the Fe - iron). So, as soon as both of the metal into the electrolyte (conductive path - salt solution), then immediately form a galvanic couple. The more active metal (standing left) is charged positively, while the less active - negative.
Back to our experience: the same thing happened in our glasses with solutions. Iron (Fe) is to the left than copper (Cu), so it is charged positively, rapidly oxidized. In a glass with zinc - zinc (Zn) is more active than iron. Therefore, until all the zinc will not rust, the iron will not be destroyed (which is often used for technical purposes).