

Color palette wthat is visible for people, is the narrow range of colors (colors of the rainbow), existing in nature. Color of visible spectrum concluded between wavelengths 750х10-9m (corresponding to the edge of the light in the red side) and 250х10-9m (match to the edge of the purple light).
Any item or substance have some color, that distinguish it from other similar items. This object has a property to absorb and reflect light. As you know, daylight is white (this light we consider when we estimate the color of the object) consists of 3 main colors: green, blue and red. Color palette consists of 7 colors of the rainbow, what were formed from these 3 colors.
We see that color of the subject, wthat reflected from its surface and that wavelength, wtat reflected from the surface of the object or light, wtat emitted by this object. Thus, the object has such color that it reflects. Other colors are absorbed by the subject and not fall on the retina of our eyes.

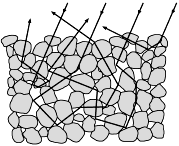

he sugar crystals are transparent, but we see it in the color, what light falls on its surface, the light repeatedly reflected in the crystal faces.
The nature of the formation of colour shades is in the structure of matter. We know from physics that the existence of Bohr model of the atom, where the electrons revolve around the atom (like planets around the sun). Each electron has a certain energy level (for ease of understanding we will compare these levels with floors of high-rise buildings). When moving from one floor to another is the release of energy - if the transition is on the lower level. If transition to a higher level, when it is absorption of energy. The release of energy is not that other, as the rays of a particular color (wavelength, wtat match the energy of made the transition). Energy absorption occurs when a light on the subject.
Complex substances, as it is known, consist of more simple substances, nterconnected at the molecular level. Some substances have a stronger chemical bond, other less strong. The stronger the link between atoms in a substance, the less intense and more light colouring. This is because the bonding electrons is "harder" to go to different energy levels ("floor"), the electrons less "free". In low bond binding electrons can leave their energy levels and transition to the adjacent levels, near of its atom and around the neighboring atom. In this is the reason for the wide spectrum of absorption of substances that have fragile chemical bond. The more heterogeneous atoms having a weak connection, the greater absorption spectrum, the more intense the color of matter, the more it black.
Why sugar white, but the crystal transparent!? The surface of the crystal is almost perfect, smooth, it is formed crystal lattice. It smoothness and the evenness can be compared to a mirror surface. As known, mirror reflects rays very well. Mirror - smooth and very thin layer of silver plates on the surface of the glass. Crystal of sugar, unlike mirrors, also has a bandwidth of light, because it faces are transparent. When light falling on the surface of the crystal, it is partially reflected from flat and smooth surface, refracted through the top face, refracted through the botton face, refracted, and go out from crystal. The light passed through the crystal, so we see crystal transparent. What happens when many crystals? In this case almost the same, but it has some changes. With all the sugar crystals are happening all the same, but when the light go out from a single crystal, it immediately goes to another, and the path of ray was repeated. So the light can travel through dozens, hundreds and thousands of crystals, and in each of the crystal will be the same. In this case, the light will receive multiple reflections from the faces of neighboring crystals, to return in the crystal again, while new crystals there will be no on his way. Thus, there is the accumulation of light energy in crystals, as it "does not pass light". That is why we see the sugar white, more precisely, in the color, that is covered light.
Refraction of light in different environments are different. Refraction depends on the refractive index of the environment, through that light passes. The refractive index equal to a mathematical ratio of the speed light in a vacuum to the speed light in the environment, which is determined by refraction. The refraction of the environment can be defined as the mathematical relation of (Sin) angle of falling to (sin) of the angle of refraction. The greater the density, the greater the refractive index. For example, the air n (air)= 1,0002926; water 1,332986; diamond 2,419; that is, if we compare the figures of refracted items through air, water and the diamond, the most distorting the image will be through a diamond.